2020-09-24 15:05
Longchi peony products related to exaggerated publicity of e-mall or suspected pyramid selling
|
[Beijing Direct News Network September 24th] (China net finance and Economics)Recently, some consumers complained to China net financial reporters that the products of Heze Longchi Biotechnology Co., Ltd. (hereinafter referred to as Longchi Biotechnology) may have problems such as false publicity, illegal production and sales. During the investigation, the reporter found that the "Haihui mall" operated by Longchi biology had also been involved in such problems as head pulling and pyramid selling. According to the national enterprise credit information publicity system, on July 1, 2020, Longchi biological was renamed as Longchi peony Industrial Co., Ltd. (hereinafter referred to as Longchi peony). Longchi peony was established on April 9, 2018. Zhang Junyu is the legal representative, executive director and general manager of the company, with a registered capital of 200 million yuan. Product may involve exaggeration The reporter searched the official website of Longchi peony and found that at present, Longchi peony has many brands such as Soule, okazi and Qingcheng peony. The official website made public its products, but did not launch a detailed introduction. 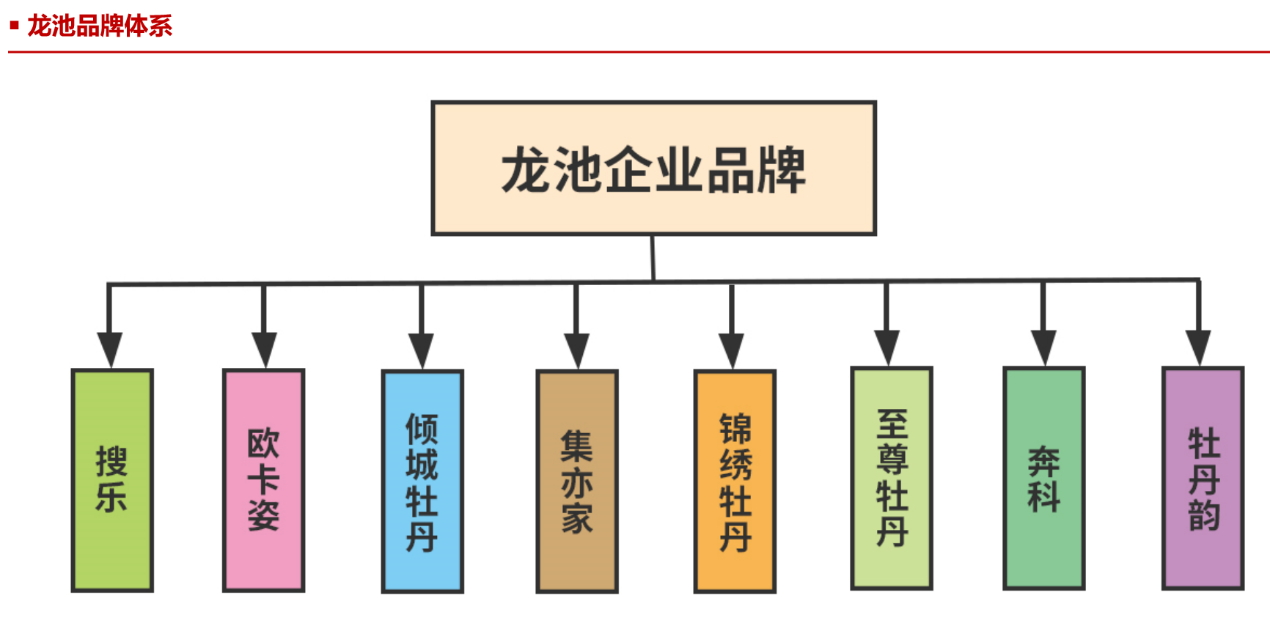 Longchi brand system Among them, the official account of the "WeChat peony" in the world, the "Peony collection" in the world, publicized the product of the cosmetics brand "Ou KASH", which was "white patent". It implied that it had whitening effect of cosmetics for whitening products. It is reported that the set contains three items: okazi pure snow penetrating muscle essence water, okazi pure snow penetrating muscle cleanser and okazi ziluhuan cream. However, the reporter found through the official website of the food and drug administration that the two kinds of cosmetics are non special cosmetics, which means that the products do not have whitening effect. Industry insiders said that the product publicity of "patent" did not indicate the patent number and patent type, which may be suspected of violating the provisions of Article 12 of the advertising law. If the advertisement involves patented products or patented methods, the patent number and patent type shall be indicated. Those who have not obtained the patent right shall not falsely claim to have obtained the patent right in the advertisement. In addition, there were two "order to correct" records in the record information of the two products mentioned above. According to the record, on March 19, 2020, the food and Drug Administration conducted a post filing inspection on Longchi biological products, and the inspection result was "ordered to correct", because "the relevant data of product safety technology were incomplete". On August 14, 2020, the food and Drug Administration once again conducted a post filing inspection on Longchi peony. The inspection result was "ordered to correct", and the reason was "incomplete data related to product safety technology".  There are two correction records in the product record of Longchi Peony The manufacturer of "ziluhuan white cream", which has the disclosure information of domestic special-purpose cosmetics, is labeled as Guangzhou Shifei Biotechnology Co., Ltd. (hereinafter referred to as Guangzhou Shifei). The approval document does not mention the information such as Longchi peony. The product category is labeled as "freckle removing". In addition, it is noted in the approval document that the State Drug Administration has not organized the review of the efficacy of the product, and this approval document is not regarded as the approval of the said efficacy of the product.
 Record information of ziluhuan white cream Some people in the industry said that, according to the provisions of Article 16 of the "Regulations on the work of cosmetics production license", the entrusting party shall be the record holder of non special purpose cosmetics or the holder of the registration certificate of cosmetics for special purposes. Even if Longchi bio entrusts Guangzhou Shifei to produce the product, the premise is that Longchi bio must be the holder of the special certificate. The product should be produced with certificate or without special certificate, suspected of illegal production and sale. Tianyan search showed that Guangzhou Shifei had failed to report occupational hazards to the work safety supervision and management department in 2018. Guangzhou Baiyun District Administration of work safety decided to give Guangzhou Shifei a warning and impose a fine of 50000 yuan. |

